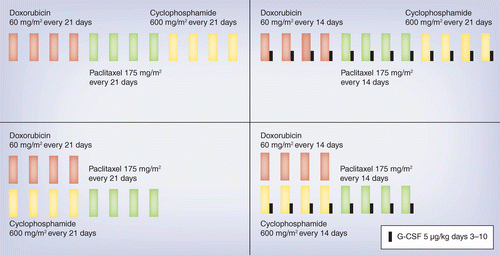
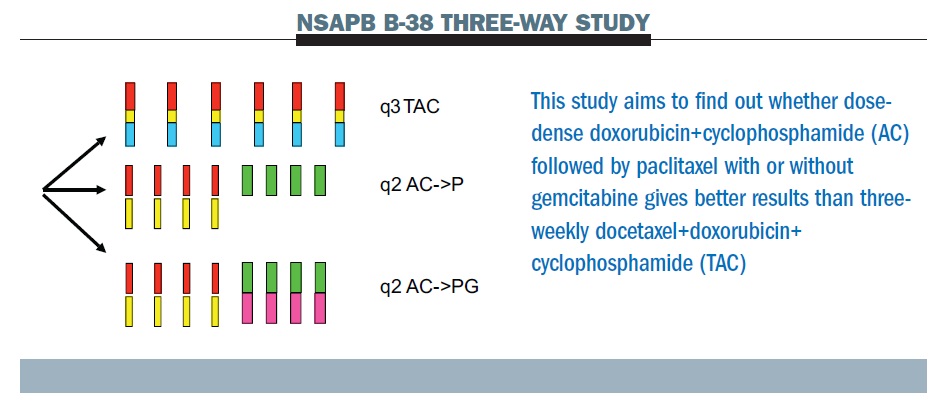
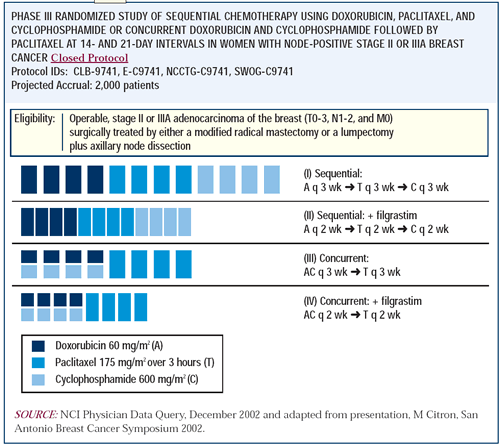
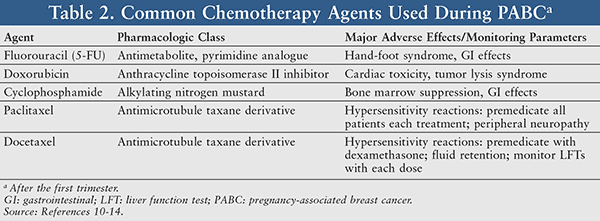
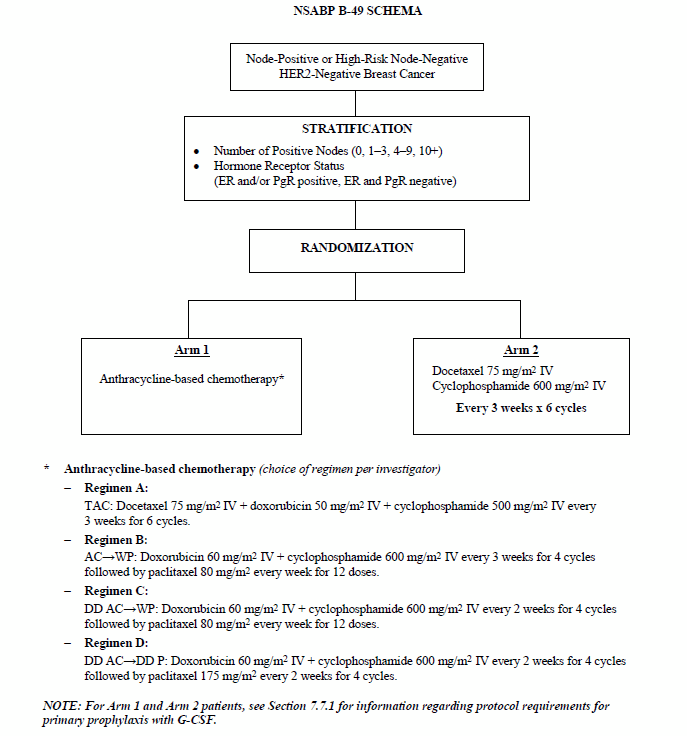
SWOG S 0800 ( NCI CDR 0000636131 ) : addition of bevacizumab to neoadjuvant nab-paclitaxel with dose-dense doxorubicin and cyclophosphamide improves pathologic complete response ( pCR ) rates in inflammatory or locally
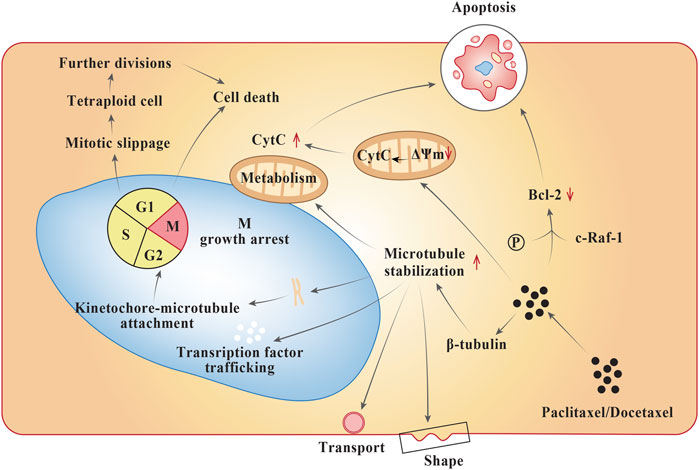
Frontiers | Platinum and Taxane Based Adjuvant and Neoadjuvant Chemotherapy in Early Triple-Negative Breast Cancer: A Narrative Review

PDF) Dose Dense Doxorubicin and Cyclophosphamide Followed by Paclitaxel (Dose Dense AC Followed by T) Regimen for Breast Cancer
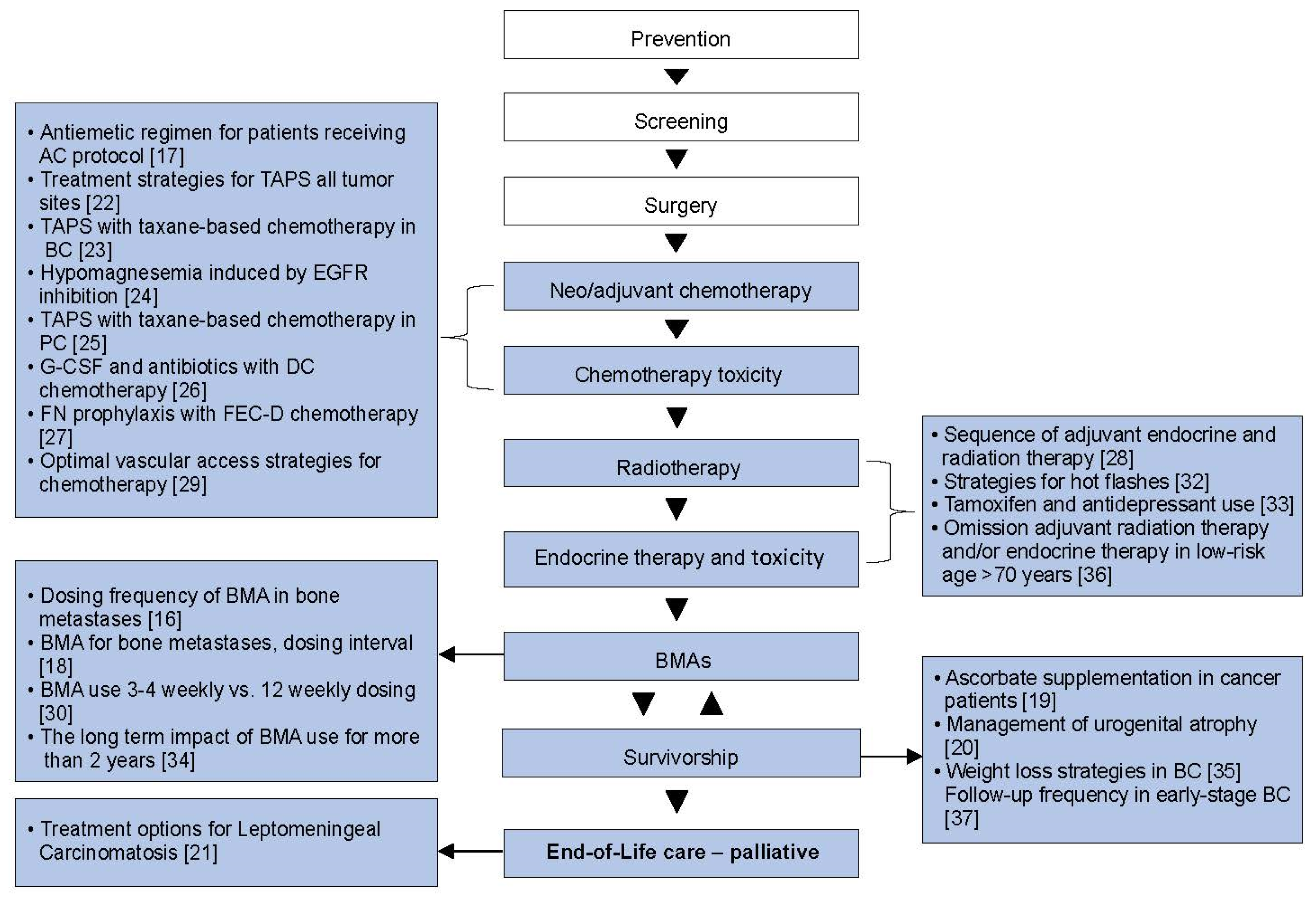
Current Oncology | Free Full-Text | Integrating Systematic Reviews into Supportive Care Trial Design: The Rethinking Clinical Trials (REaCT) Program

1607-Breast adjuvant AC (DOXOrubicin and CYCLOPHOSPHamide) dose dense followed by PACLitaxel dose de | eviQ

Sequential docetaxel as adjuvant chemotherapy for early breast cancer (TACT): an open-label, phase III, randomised controlled trial - The Lancet
![PDF] Adjuvant and neoadjuvant chemotherapy regimens in breast cancer: summary from the BSMO breast cancer task force meeting | Semantic Scholar PDF] Adjuvant and neoadjuvant chemotherapy regimens in breast cancer: summary from the BSMO breast cancer task force meeting | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2ecd93f64db58c3f9bb84457db24eb09e4422fd4/2-Figure1-1.png)
PDF] Adjuvant and neoadjuvant chemotherapy regimens in breast cancer: summary from the BSMO breast cancer task force meeting | Semantic Scholar

Anthracycline-free or short-term regimen as adjuvant chemotherapy for operable breast cancer: A phase III randomized non-inferiority trial - The Lancet Regional Health – Western Pacific

The impact of anthracyclines in intermediate and high-risk HER2-negative early breast cancer—a pooled analysis of the randomised clinical trials PlanB and SUCCESS C | British Journal of Cancer
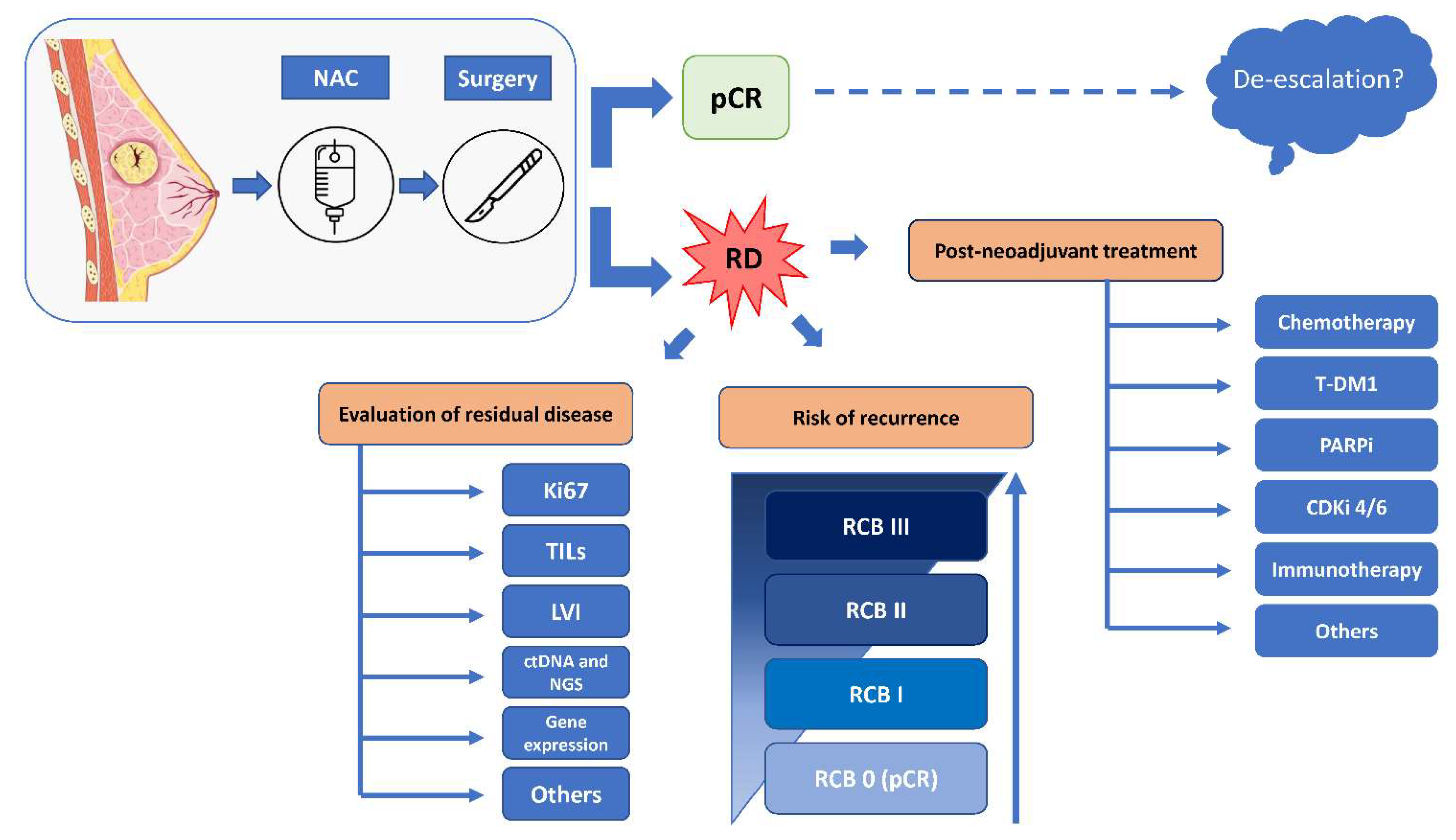
Cancers | Free Full-Text | Post-Neoadjuvant Treatment Strategies for Patients with Early Breast Cancer
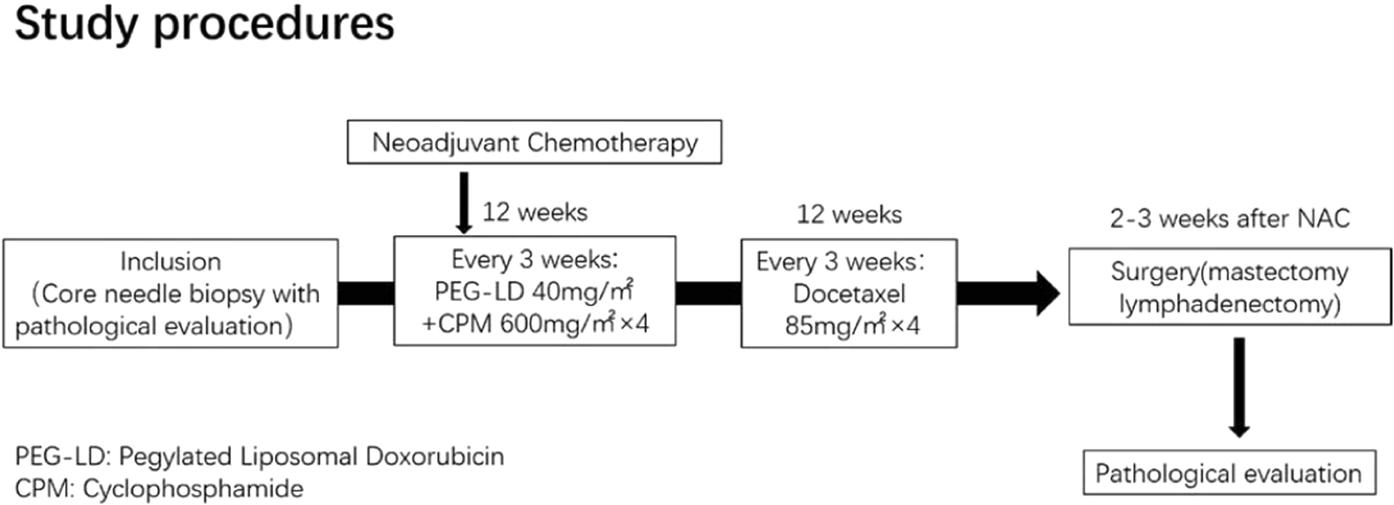
Pegylated liposomal doxorubicin plus cyclophosphamide followed by docetaxel as neoadjuvant chemotherapy in locally advanced breast cancer (registration number: ChiCTR1900023052) | Scientific Reports

CONSORT diagram for Alliance/Cancer and Leukemia Group B 49907 trial.... | Download Scientific Diagram

Timing of AC chemotherapy cycles for breast cancer patients initiating... | Download Scientific Diagram